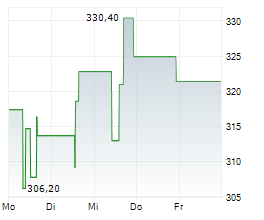
WASHINGTON (dpa-AFX) – Waters (WAT) announced that the FDA has cleared the Onclarity HPV Self-Collection Kit and approved the BD Onclarity HPV Assay with extended genotyping for at-home use. Water…
Waters Announces FDA Clearance Of Onclarity Self-Collection Kit For At-home Use
Source: Finanznachrichten.de
Read Full Story →


